Ingredients Categories
Finished Products
Finished Food Categories
- Bakery
- Beverages / Juices
- Cereal and grain products
- Confectionery
- Cooking Fats & Oils
- Dairy
- Egg products
- Fruits & Vegetables
- Infant & Baby food
- Meat, Poultry & Fish
- Nuts & Seeds products
- Other Finished Food Categories
- Pet Food
- Plant-based foods
- Prepared foods
- Sauces & Seasonings
- Sweet & Savoury Spreads
Health & Wellness Areas
Delivery Formats
Operations & Services
- 3D printing
- Analysis/ Testing
- Auditing & certifications
- Blending & mixing
- Bottling
- Certification
- Clinical Trials
- Co-packing
- Coating
- Consulting
- Contract manufacturing
- Drying
- Electrospinning
- Encapsulating
- Enzymatic processing
- Extracting
- Extrusion
- Fat structuring
- Fermentation
- Filling
- Flavour development
- Freeze Drying
- Granulating/ grinding/ milling / mixing
- High pressure processing
- Labelling
- Other Operations & Services
- Packaging
- Packet filling
- Pasturising
- Quality assurance
- Saccharification
- Sheer cell processing
- Spray drying
- Sterilisation
- Storage & Logistics
- Tabletting
- Toasting / roasting
- Ultra-high temperature processing

Guangdong Qingyunshan Pharmaceutical Co.
About
Company description
Guangdong Qingyunshan Pharmaceutical is a GMP certificate enterprise, founded in 1989, located in Guangdong, China. We have production lines for plant extraction, granules, capsules and tablets with NSF/ISO9001/ISO22000/HACCP/KOSHER/HALAL certification. Qingyunshan is a government certified High-tech Enterprise integrating R&D, production and sales of plant extract, functional foods health food and medicine. We have nearly 200 employees, including 50 R&D personnel, and 30 quality management personnel. For a long time, Qingyunshan Pharmaceutical has given full play to the advantages of experience in the field of plant extraction and separation based on the concept of "technological innovation is the core driving force of enterprise development". Qingyunshan is committed to providing high-quality products and excellent customer service.
Quick facts
| Sales markets | Africa; Asia; Australia; Central/South America; Eastern Europe; Middle East; North America; Western Europe |
|---|---|
| Primary business activity | Manufacturer: Ingredients / Additives |
| Affiliated categories: | Egg replacer |Emulsifiers, Stabilisers, Thickeners |Other Algae More |
Recently at
Vitafoods Europe 2026
05 - 07 May 2026 Fira Barcelona Gran Via, Av. Joan Carles I, 64, 08908 L'Hospitalet de Llobregat, Barcelona, Spain We were at stand UWB18 See our Exhibitor Profile See full Exhibitor ListVitafoods Europe 2025
20 - 22 May 2025 Barcelona, Spain We were at stand 3F171 See our Exhibitor Profile See full Exhibitor ListNews about Guangdong Qingyunshan Pharmaceutical Co.
21 May 2024
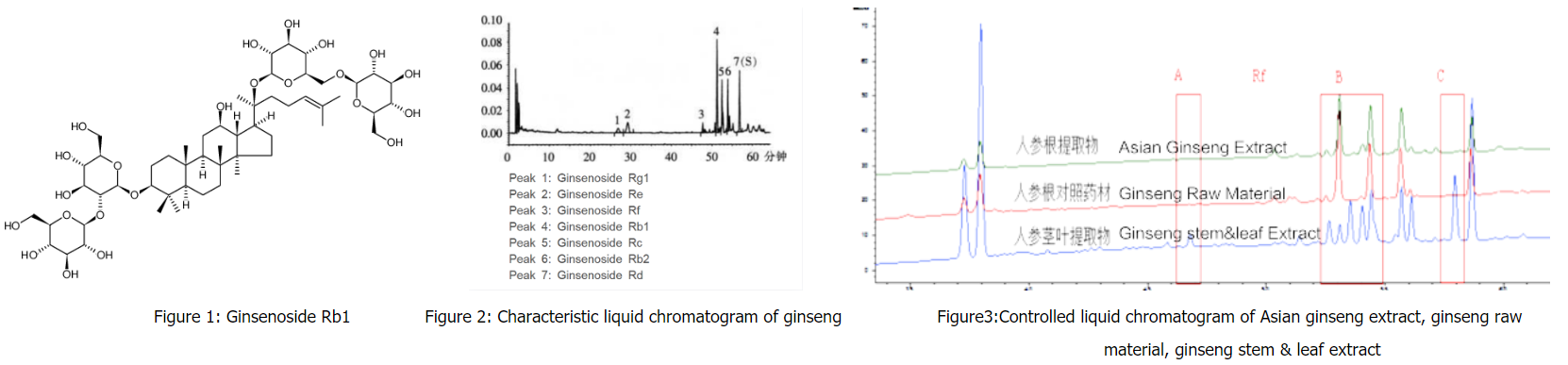
Chinese "King of Herbs" - Ginseng
Ginseng is a kind of Chinese herbal medicine with high medicinal value and has a long history in the application of clinical medicine in China.
Read more21 May 2024
The “medicine chest” of Europe - Elderberry
Elderberry (Sambucus nigra L.) is a plant belonging to the Sambucus in the Adoxaceae. It occupies an important place in the history of European herbal medicine.
Read more21 May 2024
Apple cider vinegar and weight loss
Apple Cider Vinegar And Weight Loss (herealth.com)
Read more28 Apr 2023









