Ingredients Categories
Finished Products
Finished Food Categories
- Bakery
- Beverages / Juices
- Cereal and grain products
- Confectionery
- Cooking Fats & Oils
- Dairy
- Egg products
- Fruits & Vegetables
- Infant & Baby food
- Meat, Poultry & Fish
- Nuts & Seeds products
- Other Finished Food Categories
- Pet Food
- Plant-based foods
- Prepared foods
- Sauces & Seasonings
- Sweet & Savoury Spreads
Health & Wellness Areas
Delivery Formats
Operations & Services
- 3D printing
- Analysis/ Testing
- Auditing & certifications
- Blending & mixing
- Bottling
- Certification
- Clinical Trials
- Co-packing
- Coating
- Consulting
- Contract manufacturing
- Drying
- Electrospinning
- Encapsulating
- Enzymatic processing
- Extracting
- Extrusion
- Fat structuring
- Fermentation
- Filling
- Flavour development
- Freeze Drying
- Granulating/ grinding/ milling / mixing
- High pressure processing
- Labelling
- Other Operations & Services
- Packaging
- Packet filling
- Pasturising
- Quality assurance
- Saccharification
- Sheer cell processing
- Spray drying
- Sterilisation
- Storage & Logistics
- Tabletting
- Toasting / roasting
- Ultra-high temperature processing
Sponsored Content
New UTI clinical trial
7 Mar 2024
PharmaLinea's >Your< UTI Free Sachets clinically shown to improve standard antibiotic therapy and resolve the crucial issue of recurrence
In light of the upcoming International Women’s Day, PharmaLinea has announced the publication of a new clinical study, addressing one of the most pressing global health issues for women – recurring urinary tract infections (UTIs). UTIs are highly prevalent and highly persistent. Standard antibiotic treatment, while efficient at clearing the infection, often fails to prevent relapses. In the newly published study, the addition of >Your< UTI Free Sachets to antibiotic treatment resulted in a significantly lower frequency of relapses and a significantly higher quality of life when compared to drug treatment alone. The study and product thus empower women to take control of their health and resolve an issue that previously lacked credible solutions.
The issue
UTIs affect around 400 million people yearly and about 50% of women suffer from UTIs at least once in their lifetime. The majority of patients are prescribed antibiotics, which are efficient at clearing the infection, but often fail at preventing recurrence. A staggering 30% of women affected by a UTI experience at least one recurrent infection. This substantial group of chronic UTI patients suffers a significant impact on their quality of life and has limited solutions at their disposal. The market is full of cranberry juices, teas, and commodity-based supplements that are of questionable quality and do not have clinical proof of efficacy. Doctors and pharmacists lack options they could confidently prescribe to avoid chronic urinary tract issues and chronic antibiotic use. This market gap led us to develop >Your< UTI Free Sachets in the first place and, most recently, to clinically confirm the product’s efficacy.
“Recurrent infections are not only an immediate issue for the consumer but can have larger implications. Repeating UTIs and subsequent prescriptions of antibiotics can lead to antibiotic resistance - one of the most pressing global issues. The WHO’s “Global action plan on antimicrobial resistance” includes two strategic goals: to reduce the incidence of infection and to optimize antimicrobial medicine use. With our product and the presented clinical trial, we aimed to support both those goals,“ stated Maja Orešnik, Science and Research Director at PharmaLinea.
The trial
The clinical trial aimed to evaluate the efficacy and safety of >Your< UTI Free Sachets in women with acute cystitis and their effect on the recurrence of lower UTIs. The study included 98 female patients aged 18-80 years with active acute cystitis, which was confirmed by a positive bacteriological urine test. The participants were randomized into 2 groups. Both groups received standard antibiotic therapy, while the study group additionally received >Your< UTI Free Sachets. The trial lasted 12 weeks and aimed to measure the frequency of disease relapses and assess quality of life improvements.
The study publication is accessible here.
The trial was organized and conducted by Petrovax. Uronext® is the commercial B2C brand name under which >Your< UTI Free Sachets were launched and studied.
Results
The addition of >Your< UTI Free Sachets to standard antibiotic therapy resulted in a significantly lower frequency of relapses and a significantly higher quality of life when compared to antibiotic treatment alone.
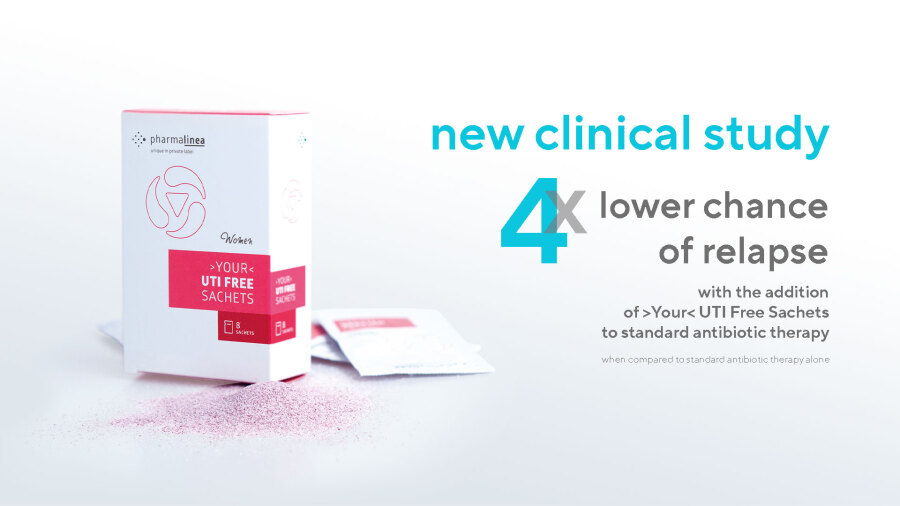
Key figures of the study group, when compared to control:
- over 4 times lower chance of relapse
- a median recurrence rate of 0
- over 20% higher improvement in quality of life
The frequency of relapses in the study group was over 4 times lower when compared to the control group. The recurrence rate in the study group was 3,7 times lower than in the control group. While the median number of recurrences per person in the control group was 1, the infection did not recur at all in the majority of the study group. The study group also reported a 20% higher improvement in quality of life during the first week of supplementation when compared to the control group. The compliance in the study group was 98%.
Trial implications
The outcome of the trial means healthcare professionals now finally have a solution they can confidently prescribe and rely on the likelihood of relapse being minimized. Women with chronic issues have gained not just a short-term remedy but a lasting resolution, leading to a substantially improved quality of life. Brand owners, in turn, are presented with a proven product they can efficiently promote through healthcare professionals, distinguishing them from competitors. Not only one but two clinical studies have now been completed on the product, adding substantial credibility.
“Already a commercial success, the product has secured the number one spot in two European markets and has been launched by brand owners in multiple markets across Europe, Asia, LATAM, and the Middle East. The new clinical data is bound to only increase the opportunity,“ said Jernej Klopčič, PharmaLinea's Business Development Director.
“It is a great example of how nutraceutical solutions can not only prevent or help with recovery but also complement traditional pharmaceutical products to improve treatment. Of course, there are other UTI supplements on the market but few have clinically supported efficacy and are thus relatively useless to urologists. Our UTI products are a unique combination of our own proprietary whole-fruit cranberry ingredient Qcran®, a clinically relevant dose of D-mannose, and microencapsulated vitamin D3, packed in a single sachet. Qcran® is one of the few if not only cranberry ingredient that is 100% whole fruit, without any sort of extraction taking place. This is achieved through a unique cold fusion production process. The resulting product retains all the beneficial properties of cranberries, including a very high fiber content, higher than any other cranberry ingredient we’ve tested. This results in the active ingredient PAC being protected, released into the bloodstream in a sustained manner, and ultimately having higher efficacy. Clinical trials are relatively rare for finished products in supplements and even more so for private label products. The investment in extensive scientific support of the product was made to empower both women and doctors with an important tool for prevention and care,“ commented PharmaLinea's Marketing Director Matevž Ambrožič.
Related news

Alvinesa Natural Ingredients announces the appointment of Olivier Lavaud as new CEO
13 May 2026
The new leadership reinforces the company’s ambition to accelerate its international growth in natural, sustainable, high value-added ingredients.
Read more
A new standard for omega-3 experience
13 Apr 2026
Omega-3 quality is no longer just about purity or concentration. Senses™ by Naturmega delivers ultra-pure, sensory-optimised omega-3 with no taste, no odour, and improved stability.
Read more
PharmaLinea’s iron supplement delivers improvements in hemoglobin comparable to ferrous sulfate with fewer side effects
23 Feb 2026
PharmaLinea, a Slovenian developer and manufacturer of private label food supplements, has announced the completion of a clinical study evaluating its finished formulation for iron deficiency.
Read more
LomaChelateX® – power in magnesium bisglycinate
12 Feb 2026
Gentle and effective: magnesium bisglycinate is setting a new standard. Through its LomaChelateX® brand, Dr. Paul Lohmann® delivers magnesium bisglycinate and a broad range of fully reacted mineral bisglycinates made in Germany.
Read more
Fucoidan leader reports record production
22 Jan 2026
The global leader in fucoidan science, Marinova Pty Ltd, reported record annual fucoidan production for 2025. The company's total production volume has more than doubled over the past three years.
Read more
India Honours Ashwagandha with Commemorative Postal Stamp at WHO Global Summit
6 Jan 2026
HYDERABAD, December 20, 2025 – India’s Prime Minister Mr. Narendra Modi, alongside WHO Director-General Dr. Tedros Adhanom Ghebreyesus, released a commemorative postal stamp honoring ashwagandha at the recently concluded Second WHO Global Traditional...
Read more
PharmaLinea delivers actionable 2026 insights at the first ever NutraLoop™ Summit
22 Dec 2025
PharmaLinea held its first-ever NutraLoop™ Summit this December. The virtual event featured 12 speakers from companies such as IQVIA, NHCO, Radicle Science, and BeyondBrands, who provided comprehensive insights across the scientific and commercia...
Read more
FrieslandCampina strengthens its global protein position with acquisition of Wisconsin Whey Protein
11 Dec 2025
Royal FrieslandCampina N.V. intends to acquire Wisconsin Whey Protein, a respected producer of whey protein isolates in the United States. Wisconsin Whey Protein will become part of FrieslandCampina Ingredients, a global leader in proteins and prebioti...
Read more
dsm-firmenich names ‘Frosted Star Anise’ as Flavor of the Year 2026, capturing a global desire for warmth and freshness
11 Dec 2025
Kaiseraugst (Switzerland), Maastricht (Netherlands), December 5, 2025 dsm-firmenich, innovators in nutrition, health, and beauty, announces ‘Frosted Star Anise’ as its Flavor of the Year for 2026. This...
Read more
Balchem Makes Strategic Investment in U.S. Food Ingredients Market with New Microencapsulation Manufacturing Facility
11 Dec 2025
Montvale, New Jersey, (10 December, 2025) – Balchem, a leading global manufacturer of specialty ingredients for human nutrition and health, is pleased to announce the development of a new, high-capacity man...
Read more
