Ingredients Categories
Finished Products
Finished Food Categories
- Bakery
- Beverages / Juices
- Cereal and grain products
- Confectionery
- Cooking Fats & Oils
- Dairy
- Egg products
- Fruits & Vegetables
- Infant & Baby food
- Meat, Poultry & Fish
- Nuts & Seeds products
- Other Finished Food Categories
- Pet Food
- Plant-based foods
- Prepared foods
- Sauces & Seasonings
- Sweet & Savoury Spreads
Health & Wellness Areas
Delivery Formats
Operations & Services
- 3D printing
- Analysis/ Testing
- Auditing & certifications
- Blending & mixing
- Bottling
- Certification
- Clinical Trials
- Co-packing
- Coating
- Consulting
- Contract manufacturing
- Drying
- Electrospinning
- Encapsulating
- Enzymatic processing
- Extracting
- Extrusion
- Fat structuring
- Fermentation
- Filling
- Flavour development
- Freeze Drying
- Granulating/ grinding/ milling / mixing
- High pressure processing
- Labelling
- Other Operations & Services
- Packaging
- Packet filling
- Pasturising
- Quality assurance
- Saccharification
- Sheer cell processing
- Spray drying
- Sterilisation
- Storage & Logistics
- Tabletting
- Toasting / roasting
- Ultra-high temperature processing

Pharmanager Development
About
Company description
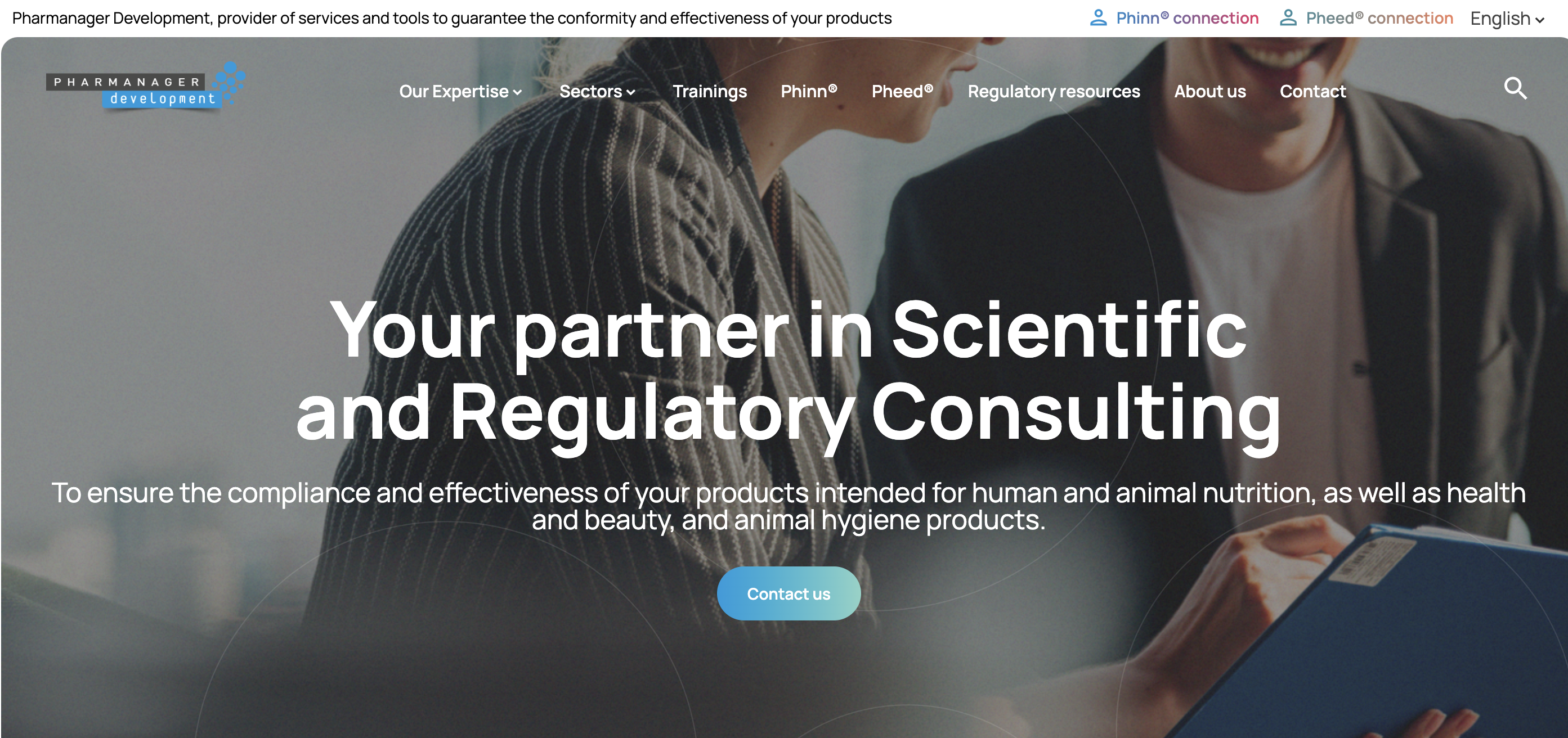
Pharmanager Development is a scientific and regulatory consulting company specialized in the nutrition and health sector, covering food products, nutraceuticals, pharmaceuticals, cosmetics, animal nutrition, animal hygiene products and biocides.
Our expertise spans 4 areas: product development, scientific substantiation, regulatory audits & procedures, training and quality audit. We assist companies in verifying product compliance and health communications, registering products when required, and preparing Novel Food dossiers for European approval. With experience in Europe and over 30 countries worldwide, we support companies in innovation and market expansion.We also provide two regulatory and scientific databases:
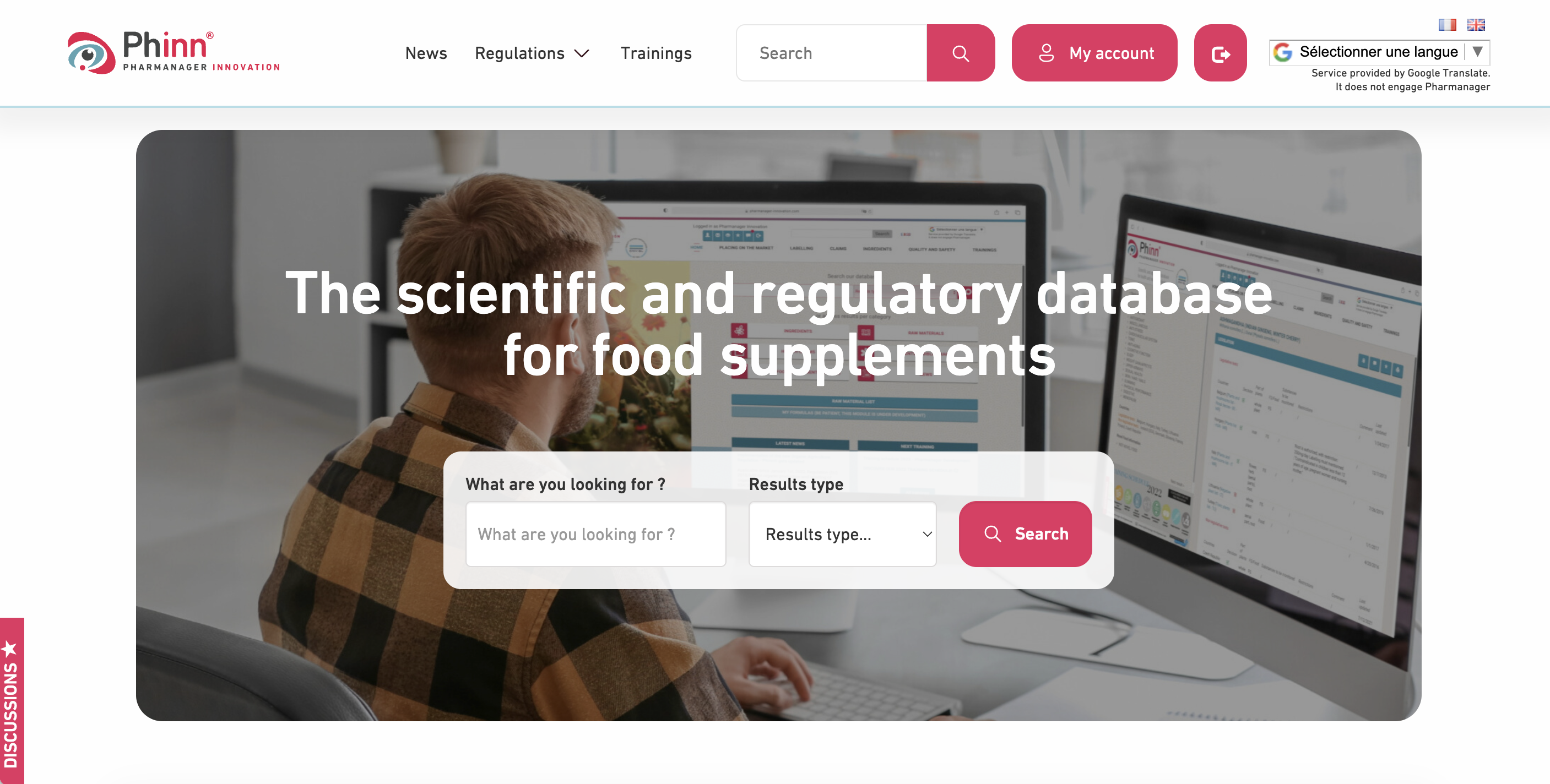
Phinn® : dedicated to food supplements:
• +230 daily users, 20+ years expertise, 97% renewal, +5000 positive clinical studies
• 2,700+ functional ingredients & additives, health/safety claims, EU regulations, 30+ third country files
• Clinical studies on health benefits, EFSA & Member States opinions
• Connection with Compl’Alim for real-time French market monitoring
• Formulas management, raw materials & suppliers database
• 1h/month personalized support (phone or email)
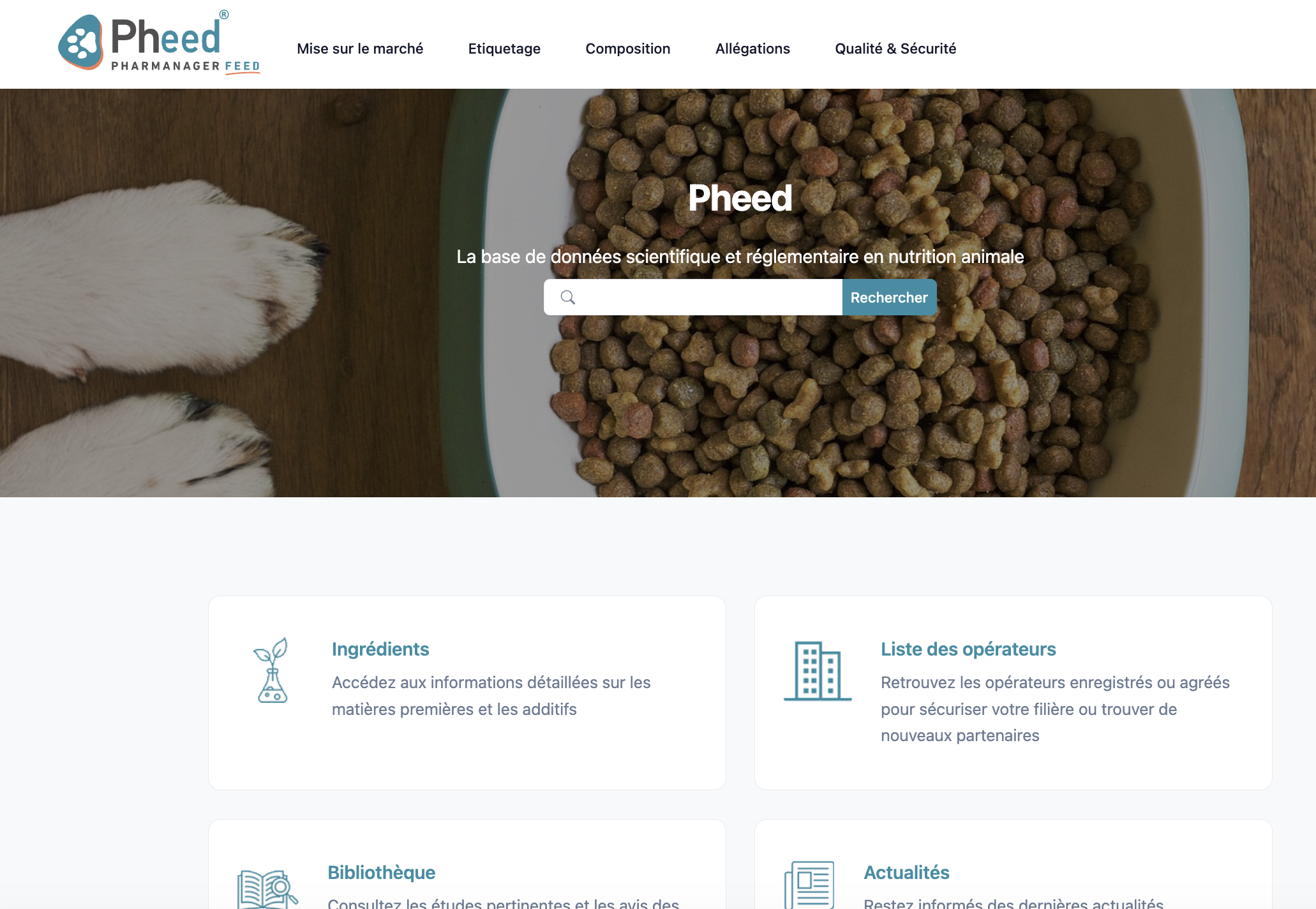
Pheed® : dedicated to animal nutrition:
• Save time, ensure compliance, foster innovation
• Centralized regulatory monitoring
• Latest scientific studies to optimize formulations
• Essential R&D data and ingredient restrictions
• Expert support to interpret regulations and address compliance challenges
• Nearly 4,500 feed materials & 1,800 additives
• Integrated list of registered operators
Quick facts
| Primary business activity | Consulting |
|---|
Recently at
Vitafoods Europe 2026
05 - 07 May 2026 Fira Barcelona Gran Via, Av. Joan Carles I, 64, 08908 L'Hospitalet de Llobregat, Barcelona, Spain We were at stand 6F109 See our Exhibitor Profile See full Exhibitor ListNews about Pharmanager Development

15 Apr 2026
Pheed® : The scientific and regulatory database for animal nutrition
Save time and ensure compliance with Pheed®, the essential scientific and regulatory database for animal nutrition professionals.
Read more15 Apr 2026
Phinn®: The scientific and regulatory database for food supplements
Phinn® is the essential database for food supplement professionals, offering compliance verification, innovation support, and a 97% renewal rate.
Read more